Barbara Davis Center for Diabetes
Peds Clinic Patient After-Hours Resources and FAQ
Learn more about what to do if you need medical advice after clinic hours and find answers to common management questions.
Diabetes and Diabetes Screening
Learn more about the symptoms, onset stages, and screening options
The EPIC Diabetes Conference is a one-day educational conference for those living with type 1 and type 2 diabetes, as well as their caregivers and healthcare providers.

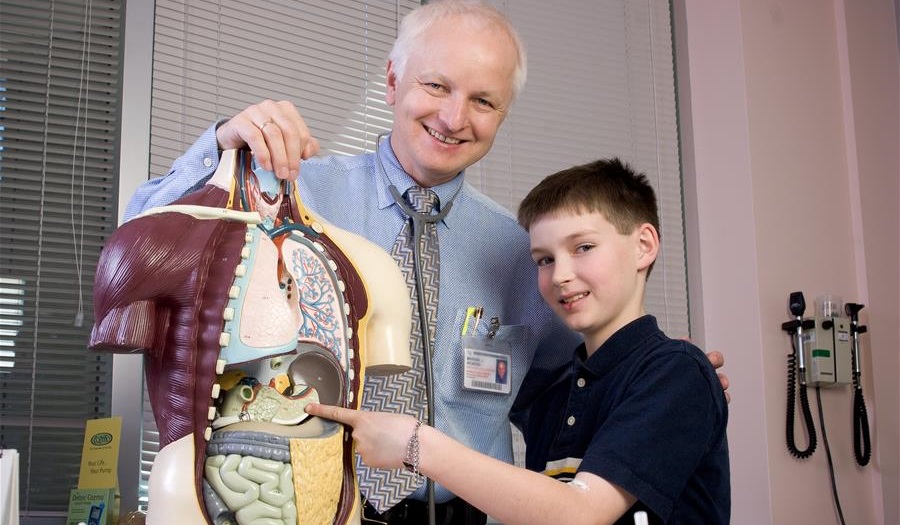
Our Mission
The Barbara Davis Center provides state-of the-art care to children and adults with type 1 diabetes and teaches our patients how to prevent or delay complications. Our research is devoted to finding prevention, cure, and most effective treatment of diabetes and associated disorders.
Marian Rewers, MD, PhD
Executive Director
What is Type 1 Diabetes?
Type 1 diabetes makes you need insulin to stay healthy. Getting diagnosed with type 1diabetes can be a surprise, so it's good to know the signs to look out for. Learn more about type 1 diabetes, what symptoms to be aware of, and when it's important to see your doctor.
Screening for Type 1 Diabetes
If someone in your family has type 1 diabetes, you and your blood-related family members have a 15-times higher chance of getting it too. Doing a simple blood test can tell you if you might develop type 1 diabetes. Most people we see in the clinic don't have a family member with type 1 diabetes, so even if you don't have a family history, it's still important to check for it. Various programs and options are available depending on your circumstances, with convenient options at the BDC and in your community.
An Infusion to Delay Type 1 Diabetes
In 2022, the FDA gave the green light for a new treatment to postpone the need to start insulin in type 1 diabetes. This treatment is for people who are currently in stage 2 of type 1 diabetes, which means they show no symptoms but have diabetes-related autoantibodies and are starting to have abnormal blood sugar levels. Our clinic evaluates patients for this infusion therapy and offers treatment as part of its state-of-the-art clinical immunotherapy program.
Main Clinic
Anschutz Medical Campus
1775 Aurora Ct. Aurora, Colorado
For directions and parking information, click here.
Outreach Clinics
For Outreach Clinic locations, click here.
Telemedicine Clinics
For Telemedicine Clinic locations, click here.
Main Phone: (303) 724-2323
Pediatric Clinic Scheduling: (303) 724-6748
Adult Clinic Scheduling: (303) 724-6755
Eye clinic Scheduling: (303) 724-6735
Additional Contact Info






