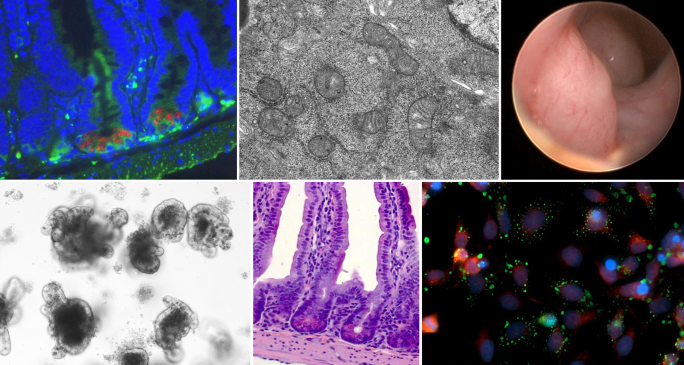
Division of Gastroenterology and Hepatology
The University of Colorado’s Division of Gastroenterology and Hepatology in the Department of Medicine, located at the Anschutz Medical Campus, is an internationally-renowned program that provides comprehensive resources for the diagnosis and therapy of diseases of the digestive tract, pancreas and liver.
Quarterly Division Newsletter
Click on the links below to view the recent happenings within the Division of Gastroenterology and Hepatology.
Division Head, Dr. Jennifer Christie's Welcome

Welcome to the University of Colorado Anschutz Medical Campus Division of Gastroenterology (GI) and Hepatology! I am excited to serve as the Division Head of this exceptional community of faculty, trainees, and staff. We are bursting with pride as we showcase the exceptional people and programs that define our division. Within our ranks, you will find experts in general gastroenterology, passionate physicians and advocates for patients with inflammatory bowel disease (IBD), highly-skilled interventional endoscopists, and dedicated professionals specializing in general and transplant hepatology. Furthermore, our basic science and clinical research programs are internationally recognized in all areas of gastroenterology and hepatology discovery.
| Contact Us |
| Gastroenterology Clinic 1635 Aurora Court Crohn's and Colitis Center Hepatology Clinic |